Paraneoplastic RTA secondary to ovarian teratoma
McKayla Muse, DO; Stillwater Medical Center;
mmuse@stillwater-medical.org
Alex Baldridge, DO; OSU Center Health Sciences;
alex.baldridge@okstate.edu
Kyrsten B Valentine, DO; OSU Center Health Sciences;
krysten.bene@okstate.edu
Matthew Payne, MD; Stillwater Medical Center;
mpayne@stillwater-medical.org
ORCID: 0009-0007-7397-6329
There are no financial sources funding this work
There are no conflicts of interest to disclose.
No financial disclosures
Introduction:
Renal tubular acidosis (RTA) should be considered in the differential diagnosis when a patient presents with persistent hypokalemia that does not respond to potassium repletion. Although the clinical presentation may not be clear, RTA often manifests with similar electrolyte abnormalities. RTA encompasses multiple subtypes, with types 1-3 which are characterized by persistent hypokalemia and non-anion gap metabolic acidosis (NAGMA).1 The specific type of RTA can be determined by the location of nephron dysfunction, either proximal or distal, and is the nature of impairment, whether it involves defective bicarbonate reabsorption or acid secretion. 1,2 Etiologies of RTA include sporadic and genetic causes, autoimmune disorders, adrenal disease, kidney disease, M-protein disorders and medications.3 Most forms of RTA are asymptomatic. Accurate identification and treatment are essential due to potential life-threatening electrolyte imbalances.
In the following case, we suspected type 1 RTA, also known as distal RTA, due to her persistent hypokalemia, non-anion gap metabolic acidosis (NAGMA), and positive urine anion gap (UAG). A well-known cause of type 1 RTA has been nephron toxicity caused by immunoglobulin protein excretion which may have played a role in the case of our patient.4 Secondary to the identification of multiple autoimmune markers and in addition to an incidental ovarian teratoma identified on imaging, a paraneoplastic phenomenon causing renal tubular dysfunction was considered. Paraneoplastic syndromes arise from tumors which secrete proteins such as hormones, peptides, or cytokines; or from an autoimmune response directed to the tumor itself.5 Although previous literature has identified a paraneoplastic relationship between ovarian teratomas and neurologic disease, there is limited information discussing these effects on the renal system.6
Case Report (methods):
We report a case of a 51-year-old female with past medical history significant for anemia and right oophorectomy, secondary to a hemorrhagic cyst in 2014. She presented to the emergency department with bilateral upper and lower extremity weakness in which symptoms began approximately 24 hours prior to arrival. Prior to her presentation, she was independent in all daily activities (ADLs). However, upon presentation she required maximum assistance with ADLs. Our patient also described 1-2 days of intermittent episodes of diarrhea. Her social history includes smoking for over 20 years, and an unknown family history due to adoption. The patient denied any prescribed medications or any routine supplements. Neurologic exam on admission was significant for decreased strength in all extremities, rated 2/5 strength. She was very tired and unable to hold a conversation. The rest of the exam was normal. Upon presentation, her potassium level was critically low at 1.5 mEq/L with EKG findings consistent with a severe hypokalemic metabolic acidosis. She had a bicarbonate level of 7 mEq/L with a corresponding pH of 7.26.
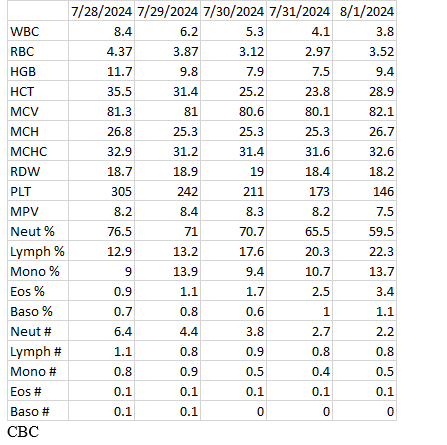
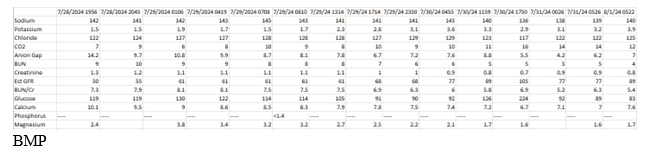
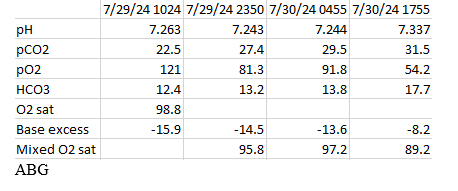
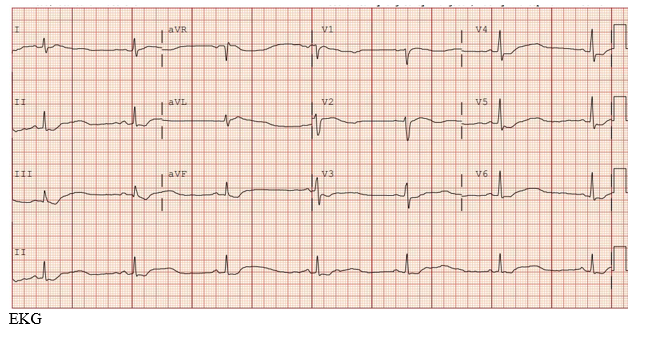
She was admitted to the Intensive Care Unit (ICU) for aggressive potassium repletion, hemodynamic monitoring and continuous lab monitoring. Within the first 24 hours, the patient received 160 mEq of potassium intravenously and 40 mEq of oral potassium. Contrary to the expected near immediate changes in her basic metabolic profile reflective of potassium supplementation, no changes were identified until approximately 18 hours after initiation of treatment. Aggressive potassium supplementation continued throughout the course of her hospitalization.
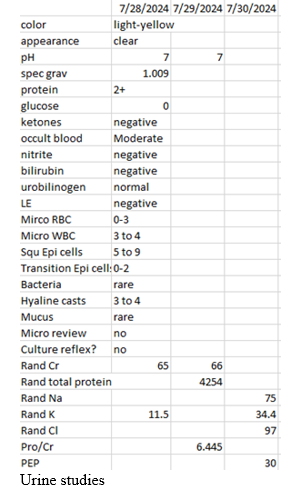
Initially, consideration was given to gastrointestinal losses as the etiology of her hypokalemia, but further investigation into her diarrheal illness did not support a volume or frequency that would be consistent with her degree of hypokalemia. Urine studies included a urine osmolality, evaluating urine sodium, urine potassium, and urine chloride. This revealed a positive UAG suggesting renal potassium loss. See figure 4. Other notable lab results revealed positive autoimmune markers including anti-ENA, anti-SS-A, and anti-SS-B antibodies.

The patient denied use of diuretics which could account for significant hypokalemia. Toxicology was unremarkable.
The constellation of history, physical exam and laboratory findings prompted us to consider autoimmune disease with an associated RTA contributing to the observed hypokalemia. Additional laboratory studies, including a cortisol level, were found to be within normal limits. A Computed Tomography (CT) of the chest, abdomen, and pelvis was completed to determine if there was an underlying hypersecreting tumor. The CT identified a large left ovarian mass consistent with a teratoma which prompted an obstetrics and gynecology consult. Based on their recommendations, the patient underwent urgent surgical removal of the mass and ovary.
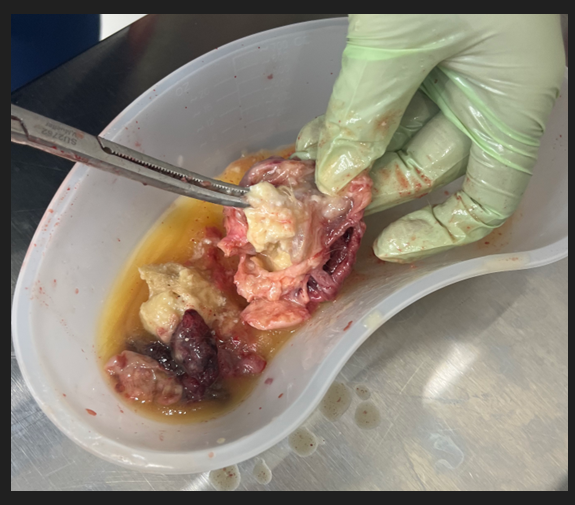
After surgery the patient was discharged on post-operative day 1 with her potassium stabilized with recommendations for close outpatient follow up.
Discussion:
Ovarian teratomas are a type of germ cell tumor that are made up of somatic tissue, typically ectoderm, endoderm, and mesoderm, which are foreign to the anatomic site they are found in. More than 95% of ovarian teratomas are mature cystic teratomas and are usually asymptomatic. Symptoms usually depend on the size of the teratoma itself. 7,8 An ovarian carcinoid neoplasm could have explained the hypokalemia caused by gastrointestinal losses from diarrhea, but this was not consistent with the volume and frequency of the patient’s presentation and not identified histopathologically.
Several studies that have revealed ovarian teratomas are associated with paraneoplastic neurological syndromes (PNS) such as anti-NMDA encephalitis (autoimmune).9,10 Based on our understanding of the underlying pathophysiology seen in paraneoplastic neurological syndromes, it may not be unreasonable to suggest a similar process affecting nephrons and resulting in an RTA. Although there are limited studies revealing an association between the proximal tubule and ovarian tumors, there is some evidence suggesting that teratomas may be associated with minimal change disease in children and SIADH in adults causing hyponatremia.11,12 Removal of the ovarian teratoma is the treatment based on the clinical picture and size.8 It is important to mention that the ovarian teratoma may have been an incidental finding, however due to the patient’s prompt recovery with stabilization of her electrolyte abnormalities after removal, it suggests that the tumor played at least a minor role in our case. By presenting this case, we hope to emphasize the potential value of further investigation and CT imaging in the detection of tumors in patients with symptoms suggestive of RTA.
References
1. Palmer, B. F., Kelepouris, E. & Clegg, D. J. RTA and management strategies: A narrative review. Adv. Ther. 38, 949–968 (2021).
2. Haque, S. K., Ariceta, G. & Batlle, D. Proximal RTA: a not so rare disorder of multiple etiologies. Nephrol. Dial. Transplant 27, 4273–4287 (2012).
3. UpToDate. https://www.uptodate.com/contents/etiology-and-diagnosis-of-distal-type-1-and-proximal-type-2-renal-tubular-acidosis.
4. Maldonado, J. E. et al. Fanconi syndrome in adults. A manifestation of a latent form of myeloma. Am. J. Med. 58, 354–364 (1975).
5. Pelosof, L. C. & Gerber, D. E. Paraneoplastic syndromes: an approach to diagnosis and treatment. Mayo Clin. Proc. 85, 838–854 (2010).
6. Kang, L. & Wan, C. Paraneoplastic syndrome in neuroophthalmology. J. Neurol. 269, 5272–5282 (2022).
7. Saleh, M. et al. Ovarian teratomas: clinical features, imaging findings and management. Abdom. Radiol. (NY) 46, 2293–2307 (2021).
8. UpToDate. https://www.uptodate.com/contents/ovarian-germ-cell-tumors-pathology-epidemiology-clinical-manifestations-and-diagnosis.
9. UpToDate. https://www.uptodate.com/contents/autoimmune-including-paraneoplastic-encephalitis-clinical-features-and-diagnosis.
10. Graus, F. & Dalmau, J. Paraneoplastic neurological syndromes. Curr. Opin. Neurol. 25, 795–801 (2012).
11. Alva, H. L. et al. A case report of mediastinal teratoma in a pediatric patient with nephrotic syndrome: Paraneoplastic syndrome or incidental finding? J. Pediatr. Hematol. Oncol. 45, e631–e634 (2023).
12. Mira, A. R., Brincat, M. R., Zalawadia, S., Sideris, M. & Dilley, J. Immature ovarian teratoma with gliomatosis peritonei, paraneoplastic hyponatremia and growing teratoma syndrome: a case report and literature review. Ann. Med. Surg. (Lond.) 86, 4907–4912 (2024).







