Pediatric Strabismus Clinical Trial Analysis and Reporting Statuses within the National Library of Medicine
Von Gray B.S., Oklahoma State University College of Osteopathic Medicine at Cherokee Nation, Office of Medical Student Research, Tahlequah, Oklahoma
Austin Young B.S., Oklahoma State University College of Osteopathic Medicine at Cherokee Nation, Office of Medical Student Research, Tahlequah, Oklahoma
Dawsyn Smith B.S., Oklahoma State University College of Osteopathic Medicine at Cherokee Nation, Office of Medical Student Research, Tahlequah, Oklahoma
Stephen Groves, M.D., Independent Investigator
Micah Hartwell, Ph.D. Oklahoma State University College of Osteopathic Medicine at Cherokee Nation, Office of Medical Student Research, Tahlequah, Oklahoma
Oklahoma State University Center for Health Sciences, Department of Psychiatry and Behavioral Sciences, Tulsa, Oklahoma
Corresponding Author: Von Gray, Oklahoma State University College of Osteopathic Medicine at Cherokee Nation, Office of Medical Student Research
Address: 19500 E Ross St., Tahlequah, OK 74464, United States.
Email von.gray@okstate.edu
Author Contributions: Dr. Hartwell had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: Gray, Hartwell; Acquisition, analysis, or interpretation of data: Gray, Young, Hartwell; Drafting of the manuscript: Gray, Young, Smith, Hartwell; Critical revision of the manuscript for important intellectual content: Smith, Groves; Statistical analysis: Hartwell; Study supervision: Hartwell.
Ethical Statement: This study was not subject to institutional review board oversight because it did not meet the regulatory definition of human subject research as defined in 45 CFR 46.102(d) and (f) of the Department of Health and Human Services’ Code of Federal Regulations. This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Conflicts of interest/Declarations: Dr. Hartwell has received research funding from the National Institute of Child Health and Human Development (U54HD113173), Human Resources Services Administration (U4AMC44250-01-02 and R41MC45951), and from the National Institute of Justice (2020-R2-CX-0014).
Funding: This study was not funded.
Abstract:
Background: Pediatric strabismus (PS) is one of the most common conditions for referral to an ophthalmologist, with a worldwide prevalence of nearly 6%. Untreated strabismus leads to amblyopia and irreversible decreased vision. Research through clinical trials (CTs) is necessary to advance treatment options. Our primary objective was to summarize current characteristics of PS CTs using the United States National Library of Medicine’s (NLM) Clinical Trial Database, providing an overview of current treatments. Our secondary objective was to assess rates of PS trial discontinuation.
Methods: We searched the NLM’s Clinical Trials Database (ClinicalTrials.gov) on October 8, 2024, to capture interventional CTs that included children. We collated trials by completion status and reported rates of type of strabismus assessed, intervention type, trial phase, enrollment, location, and funding source.
Results: Of 204 trials returned from the search, 77 met inclusion criteria. Of the trials, the most frequent type of strabismus was exotropia (32, 41.6%) followed by general strabismus (24, 31.2%), and esotropia (12, 15.6%). The most common interventions were surgical (33, 42.9%) and devices (22, 28.6%). Egypt had the most registered PS trials (21, 27.3%) followed by the United States (20, 26.0%). Of included trials, 3 (3.9%) had been discontinued. No significant associations were observed between trials reporting results and intervention type, type of strabismus, or targeted age range. However, there was a significant difference in enrollment among trials without results (59.4 [47.75]) and those reporting results (202 [219.0]; t = -3.06, P = 0.005).
Conclusions: Surgical interventions for PS were the most common intervention used and many completed trials lack reported results in the database. Egypt and the United States had the highest rates of registered PS CTs. Our study highlights the range of current CTs to help inform physicians and caregivers of individuals with PS.
Keywords: pediatric; strabismus; intervention; treatment; surgery; research waste
Introduction
Clinical trials (CTs) are necessary for the development of novel and efficacious treatments. Research trials have shown trial completion rates of 85%,1 although publication rates of trials, regardless of the outcome, are typically much lower. One cross-sectional analysis of the pharmacologic treatment of posttraumatic stress disorder among military veterans showed trial discontinuation rates of 20% and nonpublication rates of 28%.2 The Helsinki Accords highlight that the researcher has an ethical responsibility to publish results, regardless of the outcomes, to push science forward and better inform clinicians in their selection for patient healthcare and management.3 Additionally, there have been calls by the World Health Organization (WHO) and the World Medical Association to address the unethical and dismissive use of research funding.1 The United States White House Office of Science and Technology Policy (OSTP) will require all federally funded research results and underlying usable data to be made publicly accessible by the end of 2025.4 Currently, the Food and Drug Administration (FDA) requires completed studies registered within the Clinical Trials database to submit results within 12 months of trial completion, which has recently been enforced.5–7
Strabismus is loosely defined as an ocular misalignment that can result in decreased binocular vision, but may also be caused by binocular impairment or monocular vision impairment. Patients with strabismus are classified based on the direction of alignment of the affected eye(s), such as esotropia (one or both eyes inwardly turned), exotropia (outwardly turned), hypertropia (one eye elevated compared to the other), and hypotropia (one eye depressed).8 Furthermore, the classification includes the pathophysiologic causes of the misalignment. Such as comitant strabismus, paralytic strabismus stemming from neurogenic dysfunction, restrictive strabismus as a result of mechanical deficit within extraocular muscles, sensory strabismus secondary to acute or chronic loss of visual input, and syndromic strabismus through acquired or heritable illness.9 Untreated strabismus is associated with the development of amblyopia, a condition with potentially irreversible decreases in visual acuity and depth perception. The development of strabismus is increased in patients with a positive family history at a rate of 13-65%.10
Pediatric strabismus (PS) is one of the most common conditions for pediatric referrals to an ophthalmologist,9 carrying a global prevalence of 2-6% in the general population.10 Prevalence varies around the world, and it has been shown that white children in the United States are affected at a rate of 3% and African American children in the United States are affected at a rate of 2%.11 Strabismus itself causes many visual complications, and patients are also at an increased risk for underlying intracranial processes, ocular malignancies such as retinoblastoma,12 or major structural ocular defects. Moreover, research has shown that strabismus has a significant negative impact on socio-psychological factors, including dissatisfaction with one’s self-image, poor academic performance, social stigma with an increased difficulty in forming, and maintaining personal relationships.13 Given the prevalence of PS and the complications that may arise from it, advancing medical treatments and interventions are indicated.
The current treatment modalities for PS include eyeglasses or contact lenses, prism lenses, patching or eyedrops, eye muscle surgery, or vision therapy14,15—although vision therapy is not included in AAO preferred practice patterns.16 Bupivacaine and botulinum toxin injections into the extraocular eye muscles are also being studied.15,17 While there are many options, with advances in scientific technology and physiologic interventions, new treatments emerge regularly. Given the benefit of innovative treatments for pediatric strabismus, we first sought to characterize the breadth of interventional trials registered within the Clinical Trials Database, focusing on completed trials as well as current, ongoing trials. Secondarily, to address any research waste or unethical discontinuation of trials regarding PS, we sought to determine the rates of discontinuation among PS trials.
Methods
Using an observational study design, we searched for registered trials using ClinicalTrials.gov, an online CTs repository compiled by the United States National Library of Medicine (NLM), on October 8, 2024. This registry was chosen due to requirements of United States CTs, including prospective registration of respective studies with the United States NLM and consistent updates throughout the trial. Data relating to recruitment status, intervention type, participants, and funding are available for every trial in the registry.
Search Criteria
To perform our search, we used the advanced search function with the following criteria on ClinicalTrials.gov to identify CTs relevant to our objectives: “AREA[Study Type] EXPAND[Term] COVER[FullMatch] "Pediatric" AND AREA[ConditionSearch] (Strabismus) AND AREA[StdAge] EXPAND[Term] COVER[FullMatch]”
We filtered the search by selecting Child, which restricts the returns to all trials containing persons from birth through age 17 (which may or may not include adults). Further, ClinicalTrials.gov search scans to encompass multiple keywords related to the condition or intervention that is searched to expand the search returns. For strabismus, ClinicalTrials.gov included additional terms of squint, binocular eye movement disorder, squinting, cross-eyed, and eye deviation.
Study inclusion and exclusion criteria
To assess our primary objective, the characteristics of CTs for pediatric strabismus trials, we included interventional trials for pediatric strabismus in any phase. Trials were considered interventional if they used specialized eyeglasses such as overminus or prism lens therapy, patches, surgery, or other methods to correct strabismus. Trials were excluded if they were specific to or only included adults 18 years and older, or were observational. After the data were downloaded, trials were screened for inclusion in a masked, duplicative fashion by two authors (V.G. and A.Y.). After screening, these two authors reconciled their files, and a third author (M.H.) acted as arbiter in case of non-resolution.
To assess our secondary objective—the rates of trial discontinuation—only trials that were completed, withdrawn, suspended, or terminated were included, with the latter three comprising the ‘discontinued’ category. For this objective, we excluded active trials or those in the recruiting stages.
Data extraction from clinical trials
Following the search, we downloaded the full information available for each trial, including the relevant items for this study: the trial’s status, phase, location, design, type of intervention, outcome measures, funding source, and whether the results are reported. We coded location as United States-based or non-United States. Another data point used in the study was trial design— whether the study used randomized selection or masking.
Categories included in this study were esophoria, esotropia, exotropia, hypertropia, strabismic amblyopia, and general strabismus. We categorized intervention types as: exercise, drug, surgery, or device, with the device category encompassing glasses, patches, prism lenses, and devices such as virtual reality therapy, and the exercise category encompassing vision therapy. Treatments were further categorized as eyeglasses, overminus lenses, controlled intermittent alternate occlusion glasses, patching, contact lenses, prism lenses, drugs (bupivacaine and botulinum toxin injections), vision therapy, or eye muscle surgery.
Analyses
For our primary objective, we reported the total number of articles returned from the search, the number of excluded studies along with reasons for exclusion, and the number of trials included in our analysis. Next, we summarized and reported the characteristics of PS trials, including the number of participants enrolled, trial phase, state or country (if outside the United States) the study took place, trial design, type of intervention, and funding source.
Our second objective—an assessment of completed and discontinued trials—excluded trials that were still active or recruiting at the time of the search. We first reported the rates at which trials were discontinued. We planned analyses to assess potential associations between trial completion/discontinuation and study characteristics listed above using Chi-Square tests; however, low discontinuation rates prevented them from being carried out. Next, among the completed trials, we reported the frequency at which they had results reported within the Clinical Trials database. We again used Chi-Square testing to measure associations between study characteristics and reporting of results. Analyses were conducted using Stata 16.1 (StataCorp, LLC., College Station, TX), and alpha was set at 0.05 for all analyses. This study was not subject to institutional review board oversight because it did not meet the regulatory definition of human subject research as defined in 45 CFR 46.102(d) and (f) of the Department of Health and Human Services’ Code of Federal Regulations.
Results
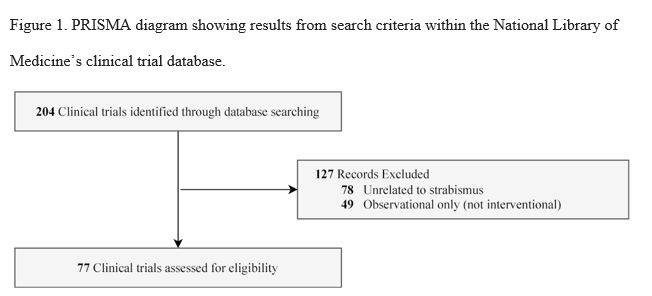
The initial data collection included 204 CTs as shown in Figure 1. No trials were excluded for lack of pediatric inclusion. Of the trials evaluated, 127 studies were excluded for the following reasons: 78 trials were excluded for being unrelated to strabismus, and 49 trials were determined to be observational only and thus excluded. In short, 77 trials met our primary objective inclusion criteria.
Study Characteristics
Of the 77 trials included in our sample, most interventions were geared toward exotropia specifically (32; 41%), with other trials treating strabismus in general (24; 31%), esotropia (12; 16%), strabismic amblyopia (7; 9%), esophoria (1; 1%), or hypertropia (1; 1%; Table 1). The interventions were sorted into five main categories: surgery (33; 43%), device (22; 29%), exercise (13; 17%), drug (8; 10%), and current standard of care treatments (1; 1%).Of the trials, 83% (64) did not provide a trial phase, leaving only 13 trials having the trial phase listed, with 1% (1) in phase 4, 8% (6) in phase 3, 4% (3) in phase 2/phase 3, 1% (1) in phase 1/phase 2, and 3% (2) in phase 1.
From the 77 pediatric strabismus trials included in our sample, 43% (33) were eye muscle surgery, 23% (18) vision therapy, 9% (7) drugs, 7% (5) patching, 5% (4) eyeglasses, 3% (2) overminus lens therapy, 3% (2) controlled intermittent alternate occlusion glasses, 3% (2) prism lenses, 1% (1) listed as treating via standard of care, 1% (1) a combination of patching and eyeglasses, 1% (1) a combination of patching and vision therapy, and 1% (1) a combination of drugs and patching.
Among all trials, 26% (20) were conducted in the United States with other trials conducted in Azerbaijan, Canada, China, Egypt, France, Germany, India, Iran, Israel, Japan, Jordan, Netherlands, Oman, Pakistan, Qatar, Singapore, South Korea, Spain, Switzerland, Taiwan, Turkey, and the United Kingdom. Of the trials assessing efficacy of eye muscle surgery, 55% (18) were in Egypt, 12% (4) in Iran, 9% (3) in China, 6% (2) in South Korea, 3% (1) in Germany, 3% (1) in Spain, 3% (1) in the United States, 3% (1) in Oman, 3% (1) in Pakistan, and 3% (1) in Canada.
Table 1. Characteristics of clinical trials for pediatric strabismus registered in the National
Library of Medicine's Clinical Trials Database.
Library of Medicine's Clinical Trials Database.
| Status |
Active/..
No (%)
|
Completed
No (%)
|
Discount..
No (%)
|
Unknown
No (%)
|
Total
No (%)
|
|
| Type of strabismus in study |
|
|
|
|
|
| Esophoria/Esotropia |
4 (5.19) |
7 (9.09) |
0 (0) |
2 (2.60) |
13 (16.88) |
| Exotropia |
8 (10.39) |
12 (15.58) |
2 (2.6) |
10 (12.99) |
32 (41.56) |
| Hypertropia |
1 (1.30) |
0 (0) |
0 (0) |
0 (0) |
1 (1.30) |
| Strabismic
Amblyopia |
1 (1.30) |
3 (3.90) |
1 (1.30) |
2 (2.60) |
7 (9.09) |
| Strabismus (General) |
3 (3.90) |
11 (14.29) |
0 (0) |
10 (12.99) |
24 (31.17) |
| Intervention Strategy |
|
|
|
|
|
| Device |
7 (9.09) |
6 (7.79) |
2 (2.60) |
7 (9.09) |
22 (28.57) |
| Drug |
2 (2.60) |
4 (5.19) |
0 (0) |
2 (2.60) |
8 (10.39) |
| Exercise |
4 (5.19) |
5 (6.49) |
1 (1.30) |
3 (3.90) |
13 (16.88) |
| Standard Of Care |
0 (0) |
1 (1.30) |
0 (0) |
0 (0) |
1 (1.30) |
| Surgery |
4 (5.19) |
17 (22.08) |
0 (0) |
12 (15.58) |
33 (42.86) |
| Age Group In Study |
|
|
|
|
|
| Child And Adult |
6 (7.79) |
17 (22.08) |
1 (1.30) |
14 (18.18) |
38 (49.35) |
| Child Only |
11 (14.29) |
16 (20.78) |
2 (2.60) |
10 (12.99) |
39 (50.65) |
| Phase Of Study |
|
|
|
|
|
| NA |
13 (16.88) |
25 (32.47) |
2 (2.60) |
17 (22.08) |
57 (74.03) |
| Phase1 |
0 (0) |
0 (0) |
1 (1.30) |
1 (1.30) |
2 (2.60) |
| Phase2 |
0 (0) |
0 (0) |
1 (1.30) |
1 (1.30) |
0 (0) |
| Phase3 |
1 (1.30) |
4 (5.19) |
0 (0) |
4 (5.19) |
9 (11.69) |
| Phase4 |
1 (1.30) |
0 (0) |
0 (0) |
0 (0) |
1 (1.30) |
|
|
|
|
|
|
|
|
|
|
|
|
| Enrollment |
|
|
|
|
|
|
114.35 |
|
|
81.55 |
91.27 |
| Mean (SD) |
(113.90) |
91.61 (122.54) |
28.33 (27.79) |
(61.61) |
(102.90) |
| Funding Source |
|
|
|
|
|
| Industry |
1 (1.30) |
1 (1.30) |
0 (0) |
1 (1.30) |
3 (3.90) |
| NIH* |
0 (0) |
3 (3.90) |
0 (0) |
1 (1.30) |
4 (5.19) |
| Other |
16 (20.78) |
29 (37.66) |
2 (2.60) |
20 (25.97) |
67 (87.01) |
| Multiple |
0 (0) |
0 (0) |
1 (1.30) |
2 (2.60) |
3 (3.90) |
| Allocation |
|
|
|
|
|
| Missing |
1 (1.30) |
2 (2.60) |
0 (0) |
1 (1.30) |
4 (5.19) |
| Non-randomized |
2 (2.60) |
10 (12.99) |
1 (1.30) |
7 (9.09) |
20 (25.97) |
| Randomized |
14 (18.18) |
21 (27.27) |
2 (2.60) |
16 (20.78) |
53 (68.83) |
|
|
|
|
|
|
| Masking |
|
|
|
|
|
| No or single |
12 (15.58) |
23 (29.87) |
2 (2.60) |
14 (18.18) |
51 (66.23) |
| Double or more |
5 (6.49) |
10 (12.99) |
1 (1.30) |
10 (12.99) |
26 (33.77) |
| CT Has Results Posted on the Clinical Trials Database |
|
|
|
|
|
| No |
17 (22.08) |
26 (33.77) |
2 (2.60) |
24 (31.17) |
69 (89.61) |
| Yes |
0 (0) |
7 (9.09) |
1 (1.30) |
0 (0) |
8 (10.39) |
*National Institutes of Health
Trial status, discontinuation, and reporting of results
For the secondary objective, 22% (17) of trials were ‘active’, ‘recruiting’, or ‘not yet recruiting’ phases and 43% (33), were completed and 31% (24) were listed as having an ‘unknown’ study status on ClinicalTrials.gov, leaving 4% (3) that were discontinued.
Among the completed trials, 52% (17) were a surgical intervention, 15% (5) vision therapy, 9% (3) drugs, 6% (2) overminus lens therapy, 6% (2) prism lenses, 3% (1) eyeglasses, 3% (1) a combination of drug intervention and patching therapy, 3% (1) standard of care, and 3% (1) patching therapy (Table 2). Of the trials in the ‘active’, recruiting’, or ‘not yet recruiting’ phases, 41% (7) were vision therapy, 24% (4) surgery, 12% (2) drug interventions, 12% (2) patching therapy, 6% (1) eyeglasses, and 6% (1) a combination of patching glasses. The three discontinued trials included a combination of patching and vision therapy, controlled intermittent alternate occlusion glasses, and vision therapy. Reported reasons for trial discontinuation listed on ClinicalTrials.gov were “difficulty to keep children in randomly assigned groups”, “recruitment inadequate and investigators leaving the study institution for new positions”, and lack of funding.
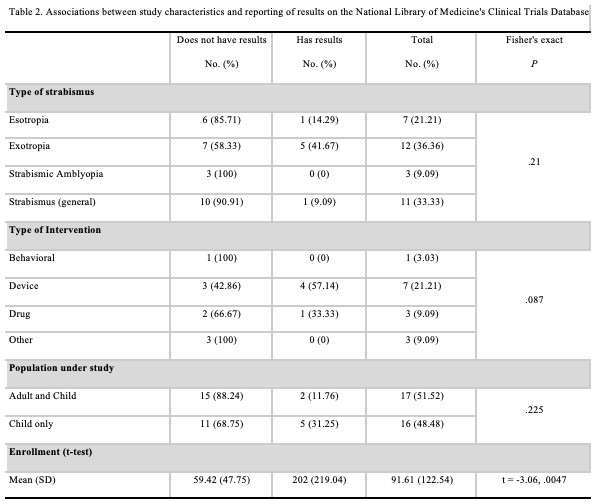
Among the 33 trials that were completed, 76% (25) lacked posting results on ClinicalTrials.gov. Only 21% (7) of completed trials had results posted, and 3% (1) had results submitted to ClinicalTrials.gov, but were not yet publicly available. Of the trials lacking results, 88% (22) were conducted outside of the United States, compared to just 12% (3) taking place within the United States. Of the three completed trials conducted within the United States that were lacking results, 100% (3) were sponsored by the National Eye Institute (NEI), with 67% (2) of the studies taking place at the NEI in Bethesda, Maryland, while 33% (1) did not provide location information. Among the completed trials with results posted, 71% (5) took place in the United States, with 14% (1) taking place in Egypt and 14% (1) taking place in Japan. The trial with results submitted but not yet publicly available took place in Egypt. All five completed trials with results reported taking place in the United States were sponsored by the Jaeb Center for Health Research. No significant associations were observed among the Fisher’s exact test results between clinical trials reporting results and intervention type, type of strabismus, or targeted age range. However, there was a significant difference in enrollment among trials without results (59.42 [SD = 47.75]) and those reporting results (202.00 [SD = 219.00]; t = -3.06, P = 0.0047; Table 2).
Discussion
Our assessment of CTs for pediatric strabismus showed that the majority included eye muscle surgery as the treatment modality. Among these surgical intervention trials, a majority were conducted in Egypt. Other treatment options were vision therapy, drugs, patching, eyeglasses, overminus lens therapy, controlled intermittent alternate occlusion glasses, prism lenses, or a combination of these. Most interventions were geared toward exotropia specifically, with other trials treating strabismus in general, esotropia, strabismic amblyopia, esophoria, or hypertropia.
Our results showed surprisingly low rates of trial discontinuation among pediatric strabismus interventions. Only three trials were discontinued, all of which were sponsored by the NEI. We recognize the fact that some trials must be discontinued or remain incomplete due to unforeseen circumstances or with regards to efficacy, feasibility of the trial, or patient safety, but it has been found that clinic trials are prematurely discontinued for legitimate reasons in only 20-30% of cases, while the large majority trial discontinuations are due to poor planning strategies.18,19 As shown, the discontinuation rate of pediatric strabismus interventions was less than 4%—lower than those found in pediatric oncology trials (15%).20 However, their study indicated that 40% of completed studies did not submit results to ClinicalTrials.gov,20 while trials for pediatric strabismus were much higher. Another study looking at the rates of discontinuation of ophthalmic trials reported rates of 5% for dry eye to 25% for age-related macular degeneration, with cataracts at 11%, cornea-related disease at 13%, diabetic macular edema/retinopathy at 16%, glaucoma at 20%, and uveitis at 10%; however, they did include trials for strabismus, nor differentiate pediatric vs. adult populations.21
Of the seven completed trials with results reported in the database, a majority were conducted by the Jaeb Center for Health Research located in Tampa, Florida, with the other two independently from GlaxoSmithKline and Cairo University. There is one completed study with results submitted but not yet posted, sponsored by Sohag University. Conversely, this leaves nearly three-quarters of the trials not having reported results—which is more than found in a study by Stewart et al for pediatric oncology trials of 40%.20 Furthermore, nearly 1 in 3 trials were listed as having an unknown study status, warranting the thought of possible discontinuation. The nonreporting of this data limits the information available, delaying the advancement of treatment options.
Active trials—either recruiting or in progress—also warrant discussion as they provide insight into the direction of pediatric strabismus research. One notable example of an ongoing trial includes the use of non-invasive, dichoptic 3D movie therapy—which may be suitable for conditions such as convergence insufficiency. It is rational to think that an intervention including patient entertainment and active participation would be a beneficial treatment strategy, especially when the treatment is directed toward a pediatric population. We appreciate the effort that researchers are putting forth in an attempt to find less invasive and more patient-friendly treatment options. Further, it is important for physicians and parents of those seeking medical intervention to be informed of the latest results from CTs of a particular condition. Ultimately, the current guidelines from the American Academy of Ophthalmology state that when conservative measures such as refractive correction and amblyopia management are unable to correct alignment, strabismus surgery is recommended as the primary treatment due to its high efficacy.16
Limitations and future research
For this study, only trials registered with ClinicalTrials.gov were included, limiting the study to those registered through the NLM. Further, those trials that may have resulted in another form of publication, for example a poster, preliminary conference report, or position paper, were not considered as published studies. While our objective was targeted toward PS, we chose search strategies to include as many trials related to strabismus interventions as possible. While this increased time spent screening, it also provided more confidence in capturing all registered CTs; however, there is a possibility that some trials were not captured. Future studies could be performed by expanding the patient population to include trials pertaining to adult populations, seeing as our search terms only had trials that included pediatrics, or be extended to assess quality of life measures or compare treatments for children versus adults. This trial is an overview of treatment options post-COVID pandemic and may not reflect pre-COVID trials, and therefore it could be of benefit to expand and perform further observational research comparing pre- to post-COVID treatments, or treatments over time. Additionally, trials for the use of adjustable sutures in those requiring reoperation of PS may be warranted given their recent use and success in the adult populations.22
Conclusion
PS encompasses multiple subtypes of strabismus, all of which are being treated using interventional therapy. Among treatment options being tested, eye muscle surgery is the most common, and about a quarter of all trials are taking place in the United States. Our data extraction demonstrated favorably low discontinuation rates and a severe lack of reporting of results from completed trials. Those performing CTs have an ethical responsibility to report results, regardless of outcome, which can improve physicians’ treatment options while also preventing future research waste.
References
1. Chalmers I, Glasziou P. Avoidable waste in the production and reporting of research evidence. Lancet. 2009;374(9683):86-89.
2. Weygandt J, Moody M, Sajjadi NB, et al. Discontinuation and nonpublication of clinical trials for the pharmacologic treatment of posttraumatic stress disorder among military veterans. J Trauma Stress. 2023;36(2):325-332.
3. Goodyear MDE, Krleza-Jeric K, Lemmens T. The declaration of Helsinki. BMJ. 2007;335(7621):624-625.
4. OSTP Issues Guidance to Make Federally Funded Research Freely Available Without Delay. The White House. August 25, 2022. Accessed October 9, 2024. https://www.whitehouse.gov/ostp/news-updates/2022/08/25/ostp-issues-guidance-to-make-federally-funded-research-freely-available-without-delay/
5. Ramachandran R, Morten CJ, Ross JS. Strengthening the FDA’s Enforcement of ClinicalTrials.gov Reporting Requirements. JAMA. Published online November 12, 2021. doi:10.1001/jama.2021.19773
6. Office of the Commissioner. Notices of Noncompliance and Civil Money Penalty Actions. September 2, 2021. Accessed December 6, 2021. https://www.fda.gov/science-research/fdas-role-clinicaltrialsgov-information/clinicaltrialsgov-notices-noncompliance-and-civil-money-penalty-actions
7. United States. Office of the Federal Register. An Act, To Amend The Federal Food, Drug, and Cosmetic Act to Revise and Extend The User-Fee Programs for Prescription Drugs,... September 27, 2007, Public Law 110-85, *.; 2008*.
8. Kanukollu VM, Sood G. Strabismus. In: StatPearls. StatPearls Publishing; 2024.
9. Ticho BH. Strabismus. Pediatr Clin North Am. 2003;50(1):173-188.
10. Çorak Eroğlu F, Oto S, Şahin Fİ, Terzi Y, Özer Kaya Ö, Tekindal MA. The role of heredity and the prevalence of strabismus in families with accommodative, partial accommodative, and infantile esotropia. Turk J Ophthalmol. 2020;50(3):143-150.
11. Friedman DS, Repka MX, Katz J, et al. Prevalence of amblyopia and strabismus in white and African American children aged 6 through 71 months the Baltimore Pediatric Eye Disease Study. Ophthalmology. 2009;116(11):2128-2134.e1-e2.
12. Pandey AN. Retinoblastoma: An overview. Saudi J Ophthalmol. 2014;28(4):310-315.
13. Silva N, Castro C, Caiado F, et al. Evaluation of functional vision and eye-related quality of life in children with strabismus. Clin Ophthalmol. 2022;16:803-813.
14. Boyd K. Strabismus in Children. American Academy of Ophthalmology. July 25, 2023. Accessed November 6, 2024. https://www.aao.org/eye-health/diseases/strabismus-in-children
15. Strabismus (crossed eyes). Accessed November 6, 2024. https://www.aoa.org/healthy-eyes/eye-and-vision-conditions/strabismus?sso=y
16. Sprunger DT, Lambert SR, Hercinovic A, et al. Esotropia and exotropia preferred practice pattern®. Ophthalmology. 2023;130(3):P179-P221.
17. Gómez de Liaño R. The use of Botulinum Toxin in strabismus treatment. J Binocul Vis Ocul Motil. 2019;69(2):51-60.
18. Psaty BM, Rennie D. Stopping medical research to save money: a broken pact with researchers and patients: A broken pact with researchers and patients. JAMA. 2003;289(16):2128-2131.
19. Lièvre M, Ménard J, Bruckert E, et al. Premature discontinuation of clinical trial for reasons not related to efficacy, safety, or feasibility. BMJ. 2001;322(7286):603-605.
20. Stewart C, McPherson K, Whitaker LA, et al. Examination of reporting status of pediatric oncology trials within the national library of medicine’s trial database. Pediatr Hematol Oncol. 2023;40(8):766-777.
21. Yilmaz T, Gallagher MJ, Cordero-Coma M, et al. Discontinuation and nonpublication of interventional clinical trials conducted in ophthalmology. Can J Ophthalmol. 2020;55(1):71-75.
22. Zhang MS, Hutchinson AK, Drack AV, Cleveland J, Lambert SR. Improved ocular alignment with adjustable sutures in adults undergoing strabismus surgery. Ophthalmology. 2012;119(2):396-402.

